Endodontic Medicine
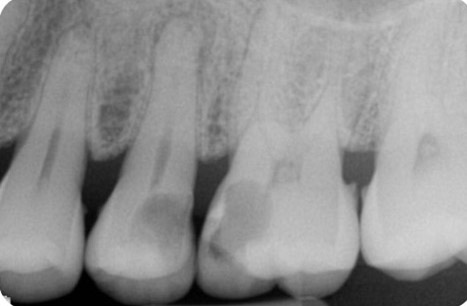
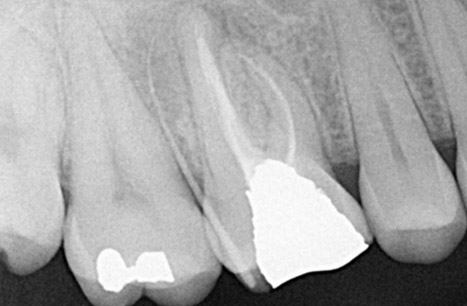
Endodontics is the study and treatment of disorders of the neurovascular tissue within the interior of teeth, including the pulp chamber and roots. These disorders occur most often when the root canal becomes infected as a result of extension of dental caries into the pulp chamber. With infection of the neurovascular tissue, necrosis can occur, and the disease process can progress to involve the entire root canal system (Figure 1). Pain, often intense, will be experienced by the affected individual. As necrotic debris becomes located at the apex of the root in the alveolar bone, the infection can extend beyond the local area to include the more coronal periodontal tissues (Figure 2). Obvious clinical swelling and suppuration can be present. The bacteria that characterize these acute lesions are Gram-negative anaerobes. These infections can be associated with systemic symptoms, including fever and malaise.
The role of oral infection/oral inflammation as risk factors for chronic diseases and disorders has generated significant attention from both the dental and medical communities. The focus of this research has been on the role of periodontal disease (periodontitis), and this field of study has been termed “Periodontal Medicine”. However, endodontic lesions can also be an important source of oral infection, and consequently inflammation, and the relationship to specific chronic diseases has been examined. This area of investigation can be referred to as “Endodontic Medicine”.
An endodontic infection that results in necrosis of the contents of the pulp chamber and canals will eventually appear on a radiograph as radiolucent area at the root apex. To illustrate the inflammatory potential of apical lesions, a study of C-reactive protein (CRP; an acute phase protein that has been associated with the systemic inflammatory burden) and interleukin-6 (IL-6; an important pro-inflammatory mediator) were evaluated in asymptomatic periapical lesions and in the periodontal ligament from teeth of unaffected patients1Garrido M, Dezerega A, Bordagaray MJ, Reyes M, Vernal R, Melgar-Rodriguez S, et al. C-reactive protein expression is up-regulated in apical lesions of endodontic origin in association with interleukin-6. J Endod. 2015;41(4):464-9.. Both types of tissue expressed CRP and IL-6, with distinctly higher levels in the tissues from periapical lesions. The authors concluded that apical lesions (apical periodontitis, AP) could contribute to persistent low-grade systemic inflammation.
A systematic review and meta-analysis evaluated the association of endodontic lesions and the concentration of inflammatory mediators in the systemic circulation2Georgiou AC, Crielaard W, Armenis I, de Vries R, van der Waal SV. Apical periodontitis is associated with elevated concentrations of inflammatory mediators in peripheral blood: A systematic review and meta-analysis. J Endod. 2019;45(11):1279-95 e3.. A total of 20 studies were included. While there was a great deal of heterogeneity in study design, the conclusion was that persons with apical periodontitis had higher concentrations of inflammatory mediators (i.e. CRP and IL-6) than controls subjects without AP. Furthermore, the complement system is a group of proteins that when cleaved help the body eliminate microorganisms. Elevated levels of certain complement components (i.e. C3) is indicative of an active infection. After endodontic treatment, the concentration of C3 in blood was reduced. The authors concluded that apical periodontitis could contribute to systemic inflammation. These lesions generally are localized and would be a source of low-grade inflammation. However, without treatment this source of inflammation could persist for lengthy periods of time.
An analysis of the leukocyte (white blood cell) concentration in blood related to endodontic infection was studied using an animal model. Three groups were established: no, one and four apical lesions3Samuel RO, Gomes-Filho JE, Azuma MM, Sumida DH, de Oliveira SHP, Chiba FY, et al. Endodontic infections increase leukocyte and lymphocyte levels in the blood. Clin Oral Investig. 2018;22(3):1395-401.. After 30 days they observed that the group with multiple lesions had higher concentrations of leukocytes as well as and the pro-inflammatory cytokine tumor necrosis factor-α (TNF-α) than either the no lesion or one lesion group. With the understanding that this is an animal model, the conclusion was that multiple endodontic lesions can alter the systemic inflammatory response, and thereby adversely affect general health.
The relationship of endodontic lesions to certain chronic disease has been examined. The focus has been on cardiovascular disease (CVD) and diabetes mellitus (DM)
Endodontic lesions and cardiovascular disease
Studies have identified an association between endodontic lesions and CVD. A review of this association was published in 2017 and identified 19 articles published between 1997 and 2016. Thirteen of the 19 studies reported a positive association, 5 of 19 did not report any association, and one study reported an inverse association4Berlin-Broner Y, Febbraio M, Levin L. Association between apical periodontitis and cardiovascular diseases: A systematic review of the literature. Int Endod J. 2017;50(9):847-59.. A number of these reports, as well as studies published after this review, are worth examining in greater detail.
An early report evaluated this relationship in the Veteran Administration Dental Longitudinal Study. A total of 708 male patients were evaluated over a 32-year period. A previous diagnosis (at baseline) of CVD was a reason for exclusion. Mean age of the participants was 47.4 years. Thirty five percent had at least one endodontic lesion and 24% were diagnosed with CVD during the follow-up period. They observed that for younger individuals, the presence of an endodontic lesion was associated with an increased risk of coronary heart disease (CHD). As an example, for every 3 years of exposure (presence of an endodontic lesion) the increased time to diagnosis of CHD was 1.4 times faster as compared to individuals who did not have an endodontic lesion5Caplan DJ, Chasen JB, Krall EA, Cai J, Kang S, Garcia RI, et al. Lesions of endodontic origin and risk of coronary heart disease. J Dent Res. 2006;85(11):996-1000..
A study of endodontic lesions and CVD risk from Austria examined the degree of calcification of the aorta (referred to as the “atherosclerotic burden”). This study was unique in that all 531 individuals (mean age of 50 years) received computed tomography of their entire body for medical reasons, and these images were then evaluated for both calcification of the aorta and apical periodontitis6Petersen J, Glassl EM, Nasseri P, Crismani A, Luger AK, Schoenherr E, et al. The association of chronic apical periodontitis and endodontic therapy with atherosclerosis. Clin Oral Investig. 2014;18(7):1813-23.. Persons with one or more endodontic lesions had nearly twice the atherosclerotic burden compared to those without a lesion (0.32 ml vs 0.17 ml; p<.05). The atherosclerotic burden was observed to increase with age and the number of untreated endodontic lesions; but not with the number of treated lesions (completed endodontic therapy). Regression analysis identified age, AP without endodontic treatment, being male and caries as determinants of the atherosclerotic burden. Endodontic therapy was seen to eliminate the adverse effect of endodontic involvement on the atherosclerotic burden.
An in-depth study of the association of endodontic lesions and CVD examined the nature of the CVD (none, stable and acute coronary syndrome as determined by angiography). Endodontic status was reported as no apical changes, a widened periodontal ligament in the apical area and an apical rarefaction. Regression analysis was used to determine associations. They concluded that endodontic lesions were an independent risk factor for CVD, with the strongest association between untreated endodontic lesions and the acute coronary syndrome7Liljestrand JM, Mantyla P, Paju S, Buhlin K, Kopra KA, Persson GR, et al. Association of endodontic lesions with coronary artery disease. J Dent Res. 2016;95(12):1358-65.. A study from Sweden demonstrated an even stronger association between apical periodontitis and CVD, with an odds ratio of 3.83 (p = 0.025)8Virtanen E, Nurmi T, Soder PO, Airila-Mansson S, Soder B, Meurman JH. Apical periodontitis associates with cardiovascular diseases: A cross-sectional study from sweden. BMC Oral Health. 2017;17(1):107..
Nevertheless, a subsequent review and meta-analysis of the association of endodontic lesions and CVD used strict criteria for inclusion into the analysis9Segura-Egea JJ, Castellanos-Cosano L, Machuca G, Lopez-Lopez J, Martin-Gonzalez J, Velasco-Ortega E, et al. Diabetes mellitus, periapical inflammation and endodontic treatment outcome. Med Oral Patol Oral Cir Bucal. 2012;17(2):e356-61.. The conclusion was that study designs and analysis of the data can be questioned, and well-designed longitudinal studies of this association were needed.
Endodontic lesions and diabetes mellitus
The relationship between periodontal disease and DM is bi-directional and has been well documented in both the dental and medical literature. The relationship between endodontic lesions and DM is not as well recognized, but there is an expanding literature on this association, and all dental clinicians should be aware of this relationship.
The association of endodontic lesions and DM has been summarized in a number published reviews. An earlier review (2012) examined both animal and clinical studies9Segura-Egea JJ, Castellanos-Cosano L, Machuca G, Lopez-Lopez J, Martin-Gonzalez J, Velasco-Ortega E, et al. Diabetes mellitus, periapical inflammation and endodontic treatment outcome. Med Oral Patol Oral Cir Bucal. 2012;17(2):e356-61.. The animal models (of both type 1 and type 2 DM) indicated that diabetes was associated more root resorption, more severe resorption of the bone at the apex of teeth and a greater inflammatory response in the pulpal tissues following pulp exposure. Subsequent clinical reviews came to similar conclusions9Segura-Egea JJ, Castellanos-Cosano L, Machuca G, Lopez-Lopez J, Martin-Gonzalez J, Velasco-Ortega E, et al. Diabetes mellitus, periapical inflammation and endodontic treatment outcome. Med Oral Patol Oral Cir Bucal. 2012;17(2):e356-61.10Segura-Egea JJ, Martin-Gonzalez J, Castellanos-Cosano L. Endodontic medicine: Connections between apical periodontitis and systemic diseases. Int Endod J. 2015;48(10):933-51.11Segura-Egea JJ, Martin-Gonzalez J, Cabanillas-Balsera D, Fouad AF, Velasco-Ortega E, Lopez-Lopez J. Association between diabetes and the prevalence of radiolucent periapical lesions in root-filled teeth: Systematic review and meta-analysis. Clin Oral Investig. 2016;20(6):1133-41.12Cintra LTA, Estrela C, Azuma MM, Queiroz IOA, Kawai T, Gomes-Filho JE. Endodontic medicine: Interrelationships among apical periodontitis, systemic disorders, and tissue responses of dental materials. Braz Oral Res. 2018;32(suppl 1):e68.13Segura-Egea JJ, Cabanillas-Balsera D, Jimenez-Sanchez MC, Martin-Gonzalez J. Endodontics and diabetes: Association versus causation. Int Endod J. 2019;52(6):790-802.14Cabanillas-Balsera D, Martin-Gonzalez J, Montero-Miralles P, Sanchez-Dominguez B, Jimenez-Sanchez MC, Segura-Egea JJ. Association between diabetes and nonretention of root filled teeth: A systematic review and meta-analysis. Int Endod J. 2019;52(3):297-306.15Perez-Losada FL, Estrugo-Devesa A, Castellanos-Cosano L, Segura-Egea JJ, Lopez-Lopez J, Velasco-Ortega E. Apical periodontitis and diabetes mellitus type 2: A systematic review and meta-analysis. J Clin Med. 2020;9(2).16Gupta A, Aggarwal V, Mehta N, Abraham D, Singh A. Diabetes mellitus and the healing of periapical lesions in root filled teeth: A systematic review and meta-analysis. Int Endod J. 2020., and reported an association of more expansive apical lesions with increased glycated hemoglobin (HbA1c) and inflammatory cytokines in blood.
A review published in 202015Perez-Losada FL, Estrugo-Devesa A, Castellanos-Cosano L, Segura-Egea JJ, Lopez-Lopez J, Velasco-Ortega E. Apical periodontitis and diabetes mellitus type 2: A systematic review and meta-analysis. J Clin Med. 2020;9(2). also included both animal and human clinical studies. Analysis of the clinical studies indicated that if a person had a diagnosis of type 2 DM, the increased risk of having a tooth with apical periodontitis was 1.17 (p=0.02), and the increased risk of a patient having at least one tooth with apical periodontitis was 1.55 (not significant).
Several individual studies are worth mentioning. Evaluation of the relationship of apical pathology to diabetes status compared patients without DM to those with disease (both metabolically well-controlled and poorly controlled patients17Smadi L. Apical periodontitis and endodontic treatment in patients with type ii diabetes mellitus: Comparative cross-sectional survey. J Contemp Dent Pract. 2017;18(5):358-62.). AP was more commonly seen in patients with DM vs. those without DM (13.5% vs 11.8%). In addition, patients with DM had a greater number of teeth that received endodontic therapy vs. the controls (4.2% vs. 1.8%; p = 0.001). Of interest, patients with poorly controlled DM had a greater prevalence of AP vs. those with well controlled DM (18.3 vs. 9.2; p = 0.001). Earlier studies have shown that patients with poorly controlled DM demonstrated larger periapical lesions compared to patients without DM. Further, when compared to controls, healing followed endodontic therapy was delayed for patients with DM18Rudranaik S, Nayak M, Babshet M. Periapical healing outcome following single visit endodontic treatment in patients with type 2 diabetes mellitus. J Clin Exp Dent. 2016;8(5):e498-e504..
Lastly, patients with DM are prone to infection with the fungus Candida albicans19Gomes CC, Guimaraes LS, Pinto LCC, Camargo G, Valente MIB, Sarquis MIM. Investigations of the prevalence and virulence of candida albicans in periodontal and endodontic lesions in diabetic and normoglycemic patients. J Appl Oral Sci. 2017;25(3):274-81.. An increase presence of C. albicans was observed in periodontal sites and samples collected from the root canal in patients with DM. These data suggest a possible role of this fungal species in the pathogenesis of both periodontal disease and endodontic infections when DM is present.
Conclusions
This review of “Endodontic Medicine” has suggested the following conclusions:
- There is an association between endodontic lesions and CVD, and endodontic lesions and DM. Cause and effect cannot be determined because most of the studies are cross-sectional.
- The mechanisms that account for the association of apical periodontitis and CVD and DM are likely similar to those that account for the association of periodontitis and CVD and DM. Specifically, periapical infection elicits a host inflammatory response, which can contribute to the systemic inflammatory burden.
- Conceptually, the relationship between AP and DM appears to be bidirectional, i.e. AP can contribute to the risk for DM via an increased systemic inflammatory burden, and DM may contribute to the severity of AP. However, longitudinal studies are required to confirm this statement.
- All patients seen in the dental office are evaluated for periodontal disease and endodontic problems. This evaluation takes on special importance for dental patients with a diagnosis of CVD and/or DM. The prevalence and severity of endodontic lesions is higher in these patients, and when present these lesions can contribute to the systemic inflammatory burden. By extension, caries prevention and treatment of existing endodontic lesions should be emphasized.
- There is no evidence that either CVD or DM contributes directly to the development of apical periodontitis. However, in poorly controlled DM xerostomia can occur, which can lead to an increased caries rate (specifically root caries). If the carious lesions progress, endodontic involvement can occur.
- Based on the published literature “endodontic lesions” should be added to the list of possible oral complications of DM. It is important to emphasize that acute infections anywhere in the body can adversely affect metabolic control in person with DM, and should be treated promptly.
Legends for Figures:
Figure 1. Radiograph demonstrating extensive caries of the distal half of the maxillary second bicuspid and mesial half of the maxillary first molar. Apical periodontitis is clearly visible for the second bicuspid and widening of the apical area of the roots of the first molar is suggested. Radiograph courtesy of Drs. Y. Berlin-Brenner and L. Levin.
Figure 2. Radiograph of a maxillary first molar demonstrating a combined endodontic-periodontal lesion. Following endodontic treatment, there is widening of the apical area of the palatal root. A gutta-percha point introduced into the gingival sulcus from the buccal surface extends to the root apex. Radiograph courtesy of Drs. Y. Berlin-Brenner and L. Levin.
- Figure 1
- Figure 2
References
- 1.Zimmerli B, Jeger F, Lussi A. Bleaching of nonvital teeth. A clinically relevant literature review. Schweiz Monatsschr Zahnmed 2010;120(4):306-20.
- 2.Brunton PA, Burke FJT, Sharif MO et al. Contemporary dental practice in the UK in 2008: Aspects of direct restorations, endodontics, and bleaching. Br Dent J 2012;212:63-7.
- 3.Demarco FF, Conde MCM, Ely C et al. Preferences on vital and nonvital tooth bleaching: A survey among dentists from a city of Southern Brazil. Braz Dent J 2013;24:527-31.
- 4.Kahler B. Present status and future directions – Managing discoloured teeth. Int Endod J 2022: Feb 21. doi: 10.1111/iej.13711.
- 5.Nutting EB, Poe GS. Chemical bleaching of discolored endodontically treated teeth. Dent Clin N Am 1967;11:655-62.
- 6.Pallarés-Serrano A, Pallarés-Serrano S, Pallarés-Serrano A, Pallarés-Sabater A. Assessment of Oxygen Expansion during Internal Bleaching with Enamel and Dentin: A Comparative In Vitro Study. Dent J 2021;9:98. https://doi.org/10.3390/dj9090098.
- 7.Estay J, Angel P, Bersezio C et al. The change of teeth color, whiteness variations and its psychosocial and self-perception effects when using low vs. high concentration bleaching gels: a one-year follow-up. BMC Oral Health 2020;20(1):255. doi: 10.1186/s12903-020-01244-x.
- 8.Haywood VB, Bergeron BE. Bleaching and the Diagnosis of Internal Resorption. Jul 24, 2018. https://decisionsindentistry.com/article/bleaching-and-the-diagnosis-of-internal-resorption/
- 9.Abbott P, Heah SY. Internal bleaching of teeth: an analysis of 255 teeth. Aust Dent J 2009;54(4):326-33. https://onlinelibrary.wiley.com/doi/epdf/10.1111/j.1834-7819.2009.01158.x.
- 10.Sakalli B, Basmaci F, Dalmizrak O. Evaluation of the penetration of intracoronal bleaching agents into the cervical region using different intraorifice barriers. BMC Oral Health 2022;22(1):266. doi: 10.1186/s12903-022-02300-4.
- 11.American Association of Endodontists Clinical Practice Committee. Use of Silver Points. AAE Position Statement. https://www.aae.org/specialty/wp-content/uploads/sites/2/2017/06/silverpointsstatement.pdf#:~:text=Silver%20points%20have%20been%20shown%20to%20corrode%20spontaneously,retreatment%20and%20replacement%20of%20the%20points%20with%20another.
- 12.Savadkouhi ST, Fazlyab M. Discoloration Potential of Endodontic Sealers: A Brief Review. Iran Endod J 2016;11(4):250-4. doi: 10.22037/iej.2016.20.
- 13.Ahmed HM, Abbott PV. Discolouration potential of endodontic procedures and materials: a review. Int Endod J 2012;45(10):883-97. doi: 10.1111/j.1365-2591.2012.02071.x.
- 14.Krastl G, Allgayer N, Lenherr P et al. Tooth discoloration induced by endodontic materials: a literature review. Dent Traumatol 2013;29(1):2-7. doi: 10.1111/j.1600-9657.2012.01141.x.
- 15.Ioannidis K, Mistakidis I, Beltes P, Karagiannis V. Spectrophotometric analysis of crown discoloration induced by MTA- and ZnOE-based sealers. J Appl Oral Sci 2013;21(2):138-44. doi: 10.1590/1678-7757201302254.
- 16.Santos LGPD, Chisini LA, Springmann CG et al. Alternative to Avoid Tooth Discoloration after Regenerative Endodontic Procedure: A Systematic Review. Braz Dent J 2018;29(5):409-18. doi: 10.1590/0103-6440201802132.
- 17.Watts A, Addy M. Tooth discoloration and staining: a review of the literature. Br Dent J 2001;190:309-16.
- 18.Pink tooth of Mummery. https://medical-dictionary.thefreedictionary.com/Pink+Tooth+of+Mummery.
- 19.Haywood VB. History, safety, and effectiveness of current bleaching techniques and applications of the nightguard vital bleaching technique. Quintessence Int 1992:23:471-88. http://www.quintpub.com/userhome/qi/qi_23_7_haywood_6.pdf
- 20.Bersezio C, Ledezma P, Mayer C et al. Effectiveness and effect of non-vital bleaching on the quality of life of patients up to 6 months post-treatment: a randomized clinical trial. Clin Oral Investig 2018;22(9):3013-9. doi: 10.1007/s00784-018-2389-y.
- 21.Popescu AD, Purcarea MV, Georgescu RV et al. Vital and Non-Vital Tooth Bleaching Procedures: A Survey Among Dentists From Romania. Rom J Oral Rehab 2021;13(3):59-71. https://www.rjor.ro/wp-content/uploads/2021/10/VITAL-AND-NON-VITAL-TOOTH-BLEACHING-PROCEDURES-A-SURVEY-AMONG-DENTISTS-FROM-ROMANIA.pdf.
- 22.Zarean P, Zarean P, Ravaghi A et al. Comparison of MTA, CEM Cement, and Biodentine as Coronal Plug during Internal Bleaching: An In Vitro Study. Int J Dent 2020;2020:8896740. doi: 10.1155/2020/8896740.
- 23.Canoglu E, Gulsahi K, Sahin C et al. Effect of bleaching agents on sealing properties of different intraorifice barriers and root filling materials. Med Oral Patol Oral Cir Bucal 2012;17 (4):e710-5. http://www.medicinaoral.com/medoralfree01/v17i4/medoralv17i4p710.pdf
- 24.Amato A, Caggiano M, Pantaleo G, Amato M. In-office and walking bleach dental treatments on endodontically-treated teeth: 25 years follow-up. Minerva Stomatol 2018;67(6):225-30.
- 25.Lim MY, Lum SOY, Poh RSC et al. An in vitro comparison of the bleaching efficacy of 35% carbamide peroxide with established intracoronal bleaching agents. Int Endod J 2004;37(7):483-8. doi: 10.1111/j.1365-2591.2004.00829.x.
- 26.Yui KCK, Rodrigues JR, Mancini MNG et al. Ex vivo evaluation of the effectiveness of bleaching agents on the shade alteration of blood-stained teeth. Int Endod J 2008;41(6):485-92. doi: 10.1111/j.1365-2591.2008.01379.x.
- 27.Warren MA, Wong M, Ingram TA III. An in vitro comparison of bleaching agents on the crowns and roots of discolored teeth. J Endod 1990;16:463-7.
- 28.Rotstein I, Mor C, Friedman S. Prognosis of intracoronal bleaching with sodium perborate preparations in vitro: 1-year study. J Endod 1993;19:10-2.
- 29.Weiger R, Kuhn A, Lost C. In vitro comparison of various types of sodium perborate used for intracoronal bleaching of discolored teeth. J Endod 1994;20:338-41.
- 30.Amato M, Scaravilli MS, Farella M, Riccitiello F. Bleaching teeth treated endodontically: long-term evaluation of a case series. J Endod 2006;32:376-8.
- 31.Abou-Rass M. Long-term prognosis of intentional endodontics and internal bleaching of tetracycline-stained teeth. Compend Contin Educ Dent 1998;19:1034-44.
- 32.Anitua E, Zabalegui B, Gil J, Gascon F. Internal bleaching of severe tetracycline discolorations: four-year clinical evaluation. Quintessence Int 1990;21(10):783-8.
- 33.Gorr S-U, Brigman HV, Anderson JC, Hirsch EB. The antimicrobial peptide DGL13K is active against drug-resistant gram-negative bacteria and sub-inhibitory concentrations stimulate bacterial growth without causing resistance. PLoS ONE 2022;17(8): e0273504. https://doi.org/10.1371/journal.pone.0273504.
- 34.Peters SM, Hill NB, Halepas S. Oral manifestations of monkeypox: A report of two cases Journal of Oral and Maxillofacial Surgery (2022). https://doi.org/10.1016/j.joms.2022.07.147.
- 35.Portalatin A. Infectious disease experts call on dentists to monitor monkeypox symptoms: 6 notes. August 15, 2022. https://www.beckersdental.com/clinical-leadership-infection-control/39124-infectious-disease-experts-call-on-dentists-to-monitor-monkeypox-symptoms-6-notes.html?origin=DentalE&utm_source=DentalE&utm_medium=email&utm_content=newsletter&oly_enc_id=1694C1316967A3F.
- 36.CDC. Monkeypox. Non-Variola Orthopoxvirus and Monkeypox Virus Laboratory Testing Data. August 17, 2022. https://www.cdc.gov/poxvirus/monkeypox/response/2022/2022-lab-test.html.
- 37.CDC. Monkeypox. Prevention. https://www.cdc.gov/poxvirus/monkeypox/prevention.html.
- 38.CDC. Monkeypox and smallpox vaccine guidance. Updated June 2, 2022. https://www.cdc.gov/poxvirus/monkeypox/clinicians/index.htm.
- 39.CDC. Monkeypox. ACAM Vaccine. https://www.cdc.gov/poxvirus/monkeypox/interim-considerations/acam2000-vaccine.html.
- 40.CDC. Guidelines for Infection Control in Dental Health-Care Settings — 2003. https://www.cdc.gov/mmwr/PDF/rr/rr5217.pdf.
- 41.CDC. Infection Prevention and Control of Monkeypox in Healthcare Settings. Updated August 11, 2022. https://www.cdc.gov/poxvirus/monkeypox/clinicians/infection-control-healthcare.html
- 42.CDC. Monkeypox. Information For Veterinarians. https://www.cdc.gov/poxvirus/monkeypox/veterinarian/index.html.
- 43.CDC. Monkeypox. Pets in the Home. https://www.cdc.gov/poxvirus/monkeypox/prevention/pets-in-homes.html.
- 44.Cheng YL, Musonda J, Cheng H, et al. Effect of surface removal following bleaching on the bond strength of enamel. BMC Oral Health 2019;19(1):50.
- 45.Monteiro D, Moreira A, Cornacchia T, Magalhães C. Evaluation of the effect of different enamel surface treatments and waiting times on the staining prevention after bleaching. J Clin Exp Dent 2017;9(5):e677-81.
- 46.Rezende M, Kapuchczinski AC, Vochikovski L, et al. Staining power of natural and artificial dyes after at-home dental bleaching. J Contemp Dent Pract 2019;20(4):424-7.


:sharpen(level=0):output(format=jpeg)/up/2023/05/Ira-Lamster-3.jpg)
:sharpen(level=0):output(format=jpeg)/up/2020/09/edodontic-medicine-2.jpg)