Introduction
Diabetes mellitus (DM) is a group of metabolic disorders characterized by elevated levels of glucose in blood. DM is associated with development of several significant clinical complications including retinopathy, nephropathy, cardiovascular/cerebrovascular disease, neuropathy, and poor wound healing. DM is also recognized as the major systemic risk factor for periodontal disease, and is associated with multiple other oral disorders, including root caries, xerostomia, Candida infection and burning mouth syndrome. Consequently, the relationship of DM and oral disease has assumed great importance for all oral health care providers and has attracted significant attention from both the dental and medical communities. To provide an update on the most recent information, a series of three essays will review important studies published between 2017 and 2021. This first essay will provide background information, and focus on periodontal disease, and endodontic complications, in association with DM.
The Prevalence of Diabetes in the United States and Across the Globe
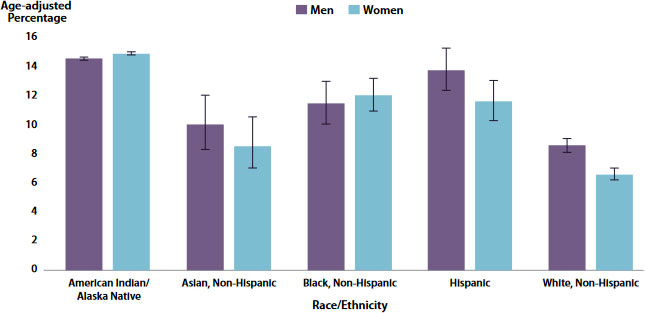
In the United States, recent prevalence data indicates that more than 10% of U.S. population have DM. Considering only adults 18 and older, that percentage is 13%. This translates to 34.1 million people. There is positive relationship of DM and age; the disease is present in 4.2% of adults 18-44 years of age. 17.5% of adults 45-64 years of age and 26.8% of adults 65 years of age and older. Further, there are differences in the prevalence of diabetes by race and ethnicity. The highest prevalence is among American Indians/Alaska Natives (14.7% of adults) and the lowest is among non-Hispanic Whites (7.5%; Figure 1).1Centers for Disease Control and Prevention. National Diabetes Statistics Report, 2020. Atlanta, GA: Centers for Disease Control and Prevention, U.S. Dept of Health and Human Services; 2020.
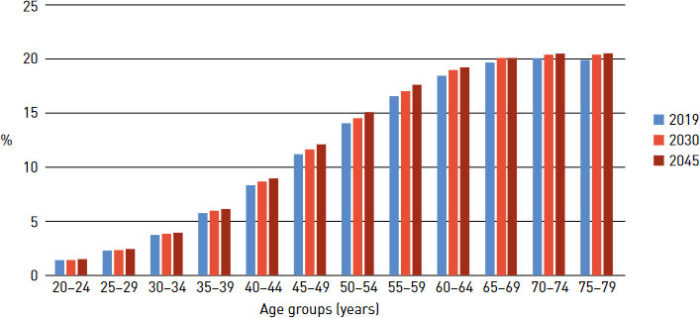
Globally, the International Diabetes Foundation estimates that in 2019 there were 463 million individuals 28 to 79 years of age with DM. The organization projects that number to increase to 700 million by 2045. Globally, the distribution of diabetes by age follows the general pattern seen for the United States, with prevalence increasing with age. This trend will remain in the future (Figure 2). Further, internationally the estimated cost of diabetes related care in 2019 was $760 billion, or 10% of the health care expenditure for adults. 2International Diabetes Federation. IDF Diabetes Atlas, 9th edn. Diabetes Facts and Figures. Brussels, Belgium: 2019. Available at: https://www.diabetesatlas.org.
Metabolic Control of Diabetes
The concentration of glucose in blood (serum glucose) was the most used method for determining metabolic control. While still used in different situations (persons with DM may monitor their fasting blood glucose level in the morning) the approach to monitoring long-term metabolic control now relies on evaluation of the concentration of glycated hemoglobin (HbA1c) in blood. This is a measurement of the amount of glucose that has attached to hemoglobin in red blood cells. Since the half-life of red blood cells is 2-3 months, this provides an assessment of the concentration of glucose in blood for the past 2-3 months. HbA1c has been shown to identify persons with DM who are at risk of developing clinical complications of the disease. Achieving good metabolic control is important in preventing or delaying the onset of diabetes complications, which are associated with significant morbidity and mortality. An evaluation of metabolic control by persons with diabetes in the United States from 1999 to 2018 revealed that following a period of improved control (1999 to 2010), the percentage of persons with good metabolic control (HbA1c <7%) declined from 57.4% to 50.5% between 2007-2010 and 2015-2018. As the population ages, this is a disturbing trend.3Fang M, Wang D, Coresh J, et al. Trends in diabetes treatment and control in U.S. adults, 1999-2018. N Engl J Med. 2021;384(23):2219-28 https://www.ncbi.nlm.nih.gov/pubmed/34107181.
These findings emphasize that individuals often have difficulty achieving ideal glycemic control over the many decades that DM may be present, and the challenges facing the healthcare system and society in general in trying to improve population health. As will be discussed in the third essay, there is an opportunity for the dental profession to be part of the answer to this dilemma. The many changes in the oral cavity associated with DM, and the identification of periodontitis as a risk factor for DM, suggest that the dental profession can assume a new role in helping to monitor patients with diagnosed DM, as well as identify their patients with previously undiagnosed pre-diabetes and DM.
What are the Oral/Dental Complications of DM?
A comprehensive review of the oral/dental complications of DM discussed the nature of the systemic complications of DM, focusing on the role of excessive inflammation and generation of reactive oxygen species (the damaging effects of molecular oxygen on cells). These pathologic conditions are particularly harmful to blood vessel (endothelial) cells and nerve cells. This accounts for the effects seen on tissues that are highly innervated and highly vascularized (i.e., the eyes, kidneys, nervous tissues). Hence, the development of retinopathy, nephropathy and neuropathy. The oral cavity is also highly innervated and highly vascularized, and many changes associated with DM are seen in the oral cavity and contiguous structures.4Verhulst MJL, Loos BG, Gerdes VEA, et al. Evaluating all potential oral complications of diabetes mellitus. Front Endocrinol (Lausanne). 2019;10:56 https://www.ncbi.nlm.nih.gov/pubmed/30962800.
This review was intended for endocrinologists, and broadly classifies complications into primary and secondary categories. The primary categories are:
1. Periodontal disease: extensive data indicates that both prevalence and incidence of periodontitis is increased in persons with diabetes, and the mechanisms appear to be similar to other microvascular complications of DM (enhanced inflammatory response). There is a positive relationship between poor glycemic control and severity of periodontitis.
2. Dental caries: an association between development of dental caries (primarily root caries) and DM has been suggested, but there are no longitudinal studies that help define cause and effect. It may be that other factors such as reduced salivary flow, and a diet rich in fermentable carbohydrates, are responsible for this association.
3. Dry mouth: this is a common complaint of patients with poorly controlled DM and is associated with reduced salivary flow. Other variables are important, including use of medications that are associated with the side effect of xerostomia.
4. Oral mucosal lesions: both Candida-associated lesions and non-Candida lesions have been identified. Candida infection is often associated with immunosuppression, and is a recognized complication of DM. This infection can take a variety of forms, including median rhomboid glossitis, angular cheilitis and denture stomatitis. There are a variety of non-Candida lesions, including ulcerations (often linked to trauma), fissured tongue and lichen planus. Mechanisms that explain the occurrence of these lesions in persons with DM have not been evaluated.
5. Oral cancer: There is an increased prevalence of pre-malignant oral lesions and oral cancer in persons with DM. This is also the case for other cancers. Mechanisms have not been defined.
6. Taste disturbances: This is a relatively rare complication, and a variety of other factors may explain this complication when it is present. These include xerostomia, and Candida infection, but may also represent a type of neuropathy.
The other (secondary) lesions include:
7. Temporomandibular joint disorders: likely associated with microvascular damage.
8. Burning Mouth Syndrome: may be a form of neuropathy, but there are other contributing factors such as reduced salivary flow.
9. Apical periodontitis related to pulpal necrosis: likely relates to the increased local and systemic inflammatory response that characterizes DM.
10. Peri-implant disease: The prevalence of peri-implant mucositis (manifesting as bleeding following probing) and peri-implantitis appear to be higher in persons with DM who have received dental implants. This again appears to be related to the increased inflammatory response in persons with DM.
The most recent published data concerning oral complications of DM has added to our understanding of many of these complications.
Periodontal Disease:
The relationship of DM to periodontal disease has been extensively studied. DM is the only systemic disease recognized as a risk factor for periodontitis (Figure 3), and if periodontitis is present, metabolic control can be adversely affected. The first statement has been repeatedly validated in the literature, and the second statement is illustrated by studies demonstrating that conservative periodontal therapy can reduce the blood level of HbA1c by 0.4% to 0.5% for 3 to 6 months following periodontal treatment.
Previously published systematic reviews have identified a bidirectional relationship between diabetes mellitus and periodontal disease. A recent systematic review examined this association by summarizing the literature to determine the relative risk of having periodontal disease if a person has DM, and having DM if a person has periodontitis.5Stohr J, Barbaresko J, Neuenschwander M, et al. Bidirectional association between periodontal disease and diabetes mellitus: a systematic review and meta-analysis of cohort studies. Sci Rep. 2021;11(1):13686 https://www.ncbi.nlm.nih.gov/pubmed/34211029. The risk of bias of the studies included in this summary was also considered. For persons with DM, the relative risk of having periodontitis was 1.24 when compared to persons without diabetes. For persons with diabetes, the relative risk of having diabetes was 1.26. These estimates may be lower than what truly exists because of the heterogeneity of the studies included in this review, primarily in terms of study design and how both DM and periodontitis were measured. Nevertheless, the authors concluded that “patients with periodontitis should be screened for diabetes mellitus, and the patients with diabetes mellitus should be informed about their higher risk of developing periodontal disease”. This study emphasizes the bidirectional nature of DM and periodontitis, and consequently the need for enhanced interprofessional practice, with close communication between medical providers caring for persons with diabetes and oral health care providers.
There is evidence that periodontal disease can be a risk factor for development of DM, and a recent study examined the data from the Atherosclerosis Risk in Communities (ARIC) study to assess this risk.6Zhang S, Philips KH, Moss K, et al. Periodontitis and Risk of Diabetes in the Atherosclerosis Risk In Communities (ARIC) Study: A BMI-modified association. J Clin Endocrinol Metab. 2021 https://www.ncbi.nlm.nih.gov/pubmed/33982107. A total of 5569 enrollees were followed for more than 19 years. Over that time, study members with advanced periodontal disease or extensive tooth loss, but also with a lower BMI (indicating that they were not obese) had an increased risk for developing DM, with adjustment for important variables including smoking and education (relative risk of 1.76 and 2.11, respectively). These data further support the systemic impact of periodontal disease. Identification of periodontal disease, and treatment to halt the progression of the disease, has beneficial effects beyond the oral cavity.
A systematic review examined the relationship of type 1 diabetes and periodontitis.7Dicembrini I, Serni L, Monami M, et al. Type 1 diabetes and periodontitis: prevalence and periodontal destruction-a systematic review. Acta Diabetol. 2020;57(12):1405-12 https://www.ncbi.nlm.nih.gov/pubmed/32318875. Previous reviews have not focused on differences between type 1 and type 2 DM. Type 1 DM is less prevalent (5-10% of cases), usually has an earlier onset (child-adolescent) than type 2 DM and has a requirement for lifelong insulin. As compared to persons in the general population, the odds ratio of having periodontitis if a person has type 1 DM was 2.5. This difference was more pronounced for persons with poor versus optimal glycemic control.
Another systematic review examined periodontal health in children with diabetes.8Zainal Abidin Z, Zainuren ZA, Noor E, et al. Periodontal health status of children and adolescents with diabetes mellitus: a systematic review and meta-analysis. Aust Dent J. 2021 https://www.ncbi.nlm.nih.gov/pubmed/33864280. A total of 11 studies were included, and clinical measures of periodontal diseases (plaque index, gingival index, clinical attachment loss and probing pocket depth) were all greater in children with DM versus age-matched children without DM. The conclusion was that when children are diagnosed with DM, evaluation of periodontal status is important to prevent development of periodontal disease.
A clinical trial looked more closely at the development of periodontal disease (both gingivitis and periodontitis) in children with type 1 diabetes.9Chakraborty P, Mukhopadhyay P, Bhattacharjee K, et al. Periodontal disease in type 1 diabetes mellitus: influence of pubertal stage and glycemic control. Endocr Pract. 2021;27(8):765-8 https://www.ncbi.nlm.nih.gov/pubmed/33486090. An analysis of adolescents with type 1 DM and healthy, age-matched subjects considering the stage of puberty, HbA1c and periodontal status. Both gingivitis and periodontitis were observed in patients with type 1 DM, and clinical parameters of periodontal disease (gingival index, plaque index, bleeding on probing and clinical attachment loss) were more severe in the type 1 DM group. In both groups, onset of puberty was associated with more pronounced periodontal disease. Using regression analysis, the stage of puberty was the most important risk factor of periodontal disease (odds ratio = 14.26) as compared to age, DM status (including duration of DM) and HbA1c. These data indicate that when evaluating the occurrence of periodontal disease in adolescents, the stage of puberty is another variable that must be considered.
Considering the bidirectional nature of DM and periodontal disease, a related question is whether a diagnosis of periodontal disease can be associated with unidentified DM. This question was reviewed in the systemic review by Stoher et al.5Stohr J, Barbaresko J, Neuenschwander M, et al. Bidirectional association between periodontal disease and diabetes mellitus: a systematic review and meta-analysis of cohort studies. Sci Rep. 2021;11(1):13686 https://www.ncbi.nlm.nih.gov/pubmed/34211029. A recent study emphasized this finding.10Heji ES, Bukhari AA, Bahammam MA, et al. Periodontal disease as a predictor of undiagnosed diabetes or prediabetes in dental patients. Eur J Dent. 2021;15(2):216-21 https://www.ncbi.nlm.nih.gov/pubmed/33285572. Adults 35 years and older who had not received a diagnosis of diabetes and had one or more risk factors for the disease were studied. About 60% of participants had advanced periodontitis. Regression analysis was used to develop a profile of who was at risk for DM. This included smoking, hypertension, a family history of DM and the percent of periodontal sites with more than 3 mm of clinical attachment loss. This algorithm identified about 70% of participants with DM. A family history of diabetes was a particularly important risk factor (odds ratio of 4.98). The authors suggested screening for DM in the dental office for patients with these risk factors.
The effect of periodontal treatment for patients with periodontitis and DM has been the basis for determining the impact of periodontal infection/inflammation on metabolic control. As noted, this treatment has been associated with 0.4%-0.5% reduction in HbA1c for 3-6 months after periodontal treatment. A confirmatory clinical study11Rapone B, Ferrara E, Corsalini M, et al. Inflammatory status and glycemic control level of patients with type 2 diabetes and periodontitis: a randomized clinical trial. Int J Environ Res Public Health. 2021;18(6) https://www.ncbi.nlm.nih.gov/pubmed/33804123. of patients with type 2 DM examined this effect. Half of the study group received conservative periodontal treatment at the start of the 6-month study, and for the other half treatment was delayed until after the study. For the initial intervention group the HbA1c values were 7.7% at baseline, 7.25% at 3 months and 7.2% at 6 months. The difference between baseline and 3 months was not statistically significant but the difference between baseline and 6 months was significant. For the delayed intervention group, HbA1c at baseline was 7.7% at baseline, 7.9% at 3 months and 7.3% at 6 months. These investigators also examined serum levels of c-reactive protein (CRP) in blood, an important indicator of systemic inflammation. At baseline, for the immediate intervention group the CRP concentration was 2.12 mg/L, 1.43 mg/L at 3 months and 1.32 mg/L at 6 months (3 and 6 months were significantly lower than baseline). For the delayed intervention group, the values were 2.12 mg/L at baseline, 2.5 mg/L at 3 months and 2.33 mg/L at 6 months. There were no statistically significant differences between these time points. The findings for HbA1c demonstrated a marked reduction after baseline for the immediate intervention group over the next 6 months. For the delayed intervention group, there was no change from baseline to 3 months, and then a decrease at 6 months. This last finding was attributed to variations in metabolic markers over time but may also have been due to the relatively small number of participants, as well as the positive effect of being in a clinical trial, even for those that are in a control group. The results for CRP are quite clear and indicate that periodontal treatment can reduce the systemic inflammatory burden.
A recent systematic review12Chen YF, Zhan Q, Wu CZ, et al. Baseline HbA1c level influences the effect of periodontal therapy on glycemic control in people with type 2 diabetes and periodontitis: a systematic review on randomized controlled trails. Diabetes Ther. 2021;12(5):1249-78 https://www.ncbi.nlm.nih.gov/pubmed/33481189. of the effect of periodontal treatment on glycemic control in persons with type 2 DM calculated a reduction in HbA1c of 0.514% at 3 months and 0.548% at 6 months. This confirmed the conclusions of earlier systematic reviews. However, they observed that differences in baseline HbA1c at baseline was the most important variable accounting for the heterogeneity. This is a clear message that baseline metabolic control should be defined for all future studies that examine the effect of periodontal treatment on metabolic control in persons with DM.
Mechanisms that Explain the DM - Periodontitis Linkage
The relationship between DM and periodontal disease is mediated by bacterial-induced inflammation, and several recent studies have examined potential underlying mechanisms to explain this association. Using a technique that evaluates the entire genome, polymorphisms of 5 genes (which are variations of a gene) were identified in persons with DM and periodontitis, but other epidemiologic variables were considered as part of this potential association including sex and smoking.13Cirelli T, Nepomuceno R, Goveia JM, et al. Association of type 2 diabetes mellitus and periodontal disease susceptibility with genome-wide association-identified risk variants in a Southeastern Brazilian population. Clin Oral Investig. 2021;25(6):3873-92 https://www.ncbi.nlm.nih.gov/pubmed/33392810. This study only examined persons in southwestern Brazil but highlights the complex nature of the genetic control of the host response in DM and periodontitis.
While it is generally agreed that the microflora in DM-associated periodontitis does not differ from that seen in periodontitis when diabetes is not present, a recent study examined the prevalence of different genera of periodontal bacteria in persons with periodontitis, with one group having a diagnosis of DM and the other without DM.14Balmasova IP, Olekhnovich EI, Klimina KM, et al. Drift of the subgingival periodontal microbiome during chronic periodontitis in type 2 diabetes mellitus patients. Pathogens. 2021;10(5) https://www.ncbi.nlm.nih.gov/pubmed/33922308. Quantitative differences were seen in the genera that were present [i.e., more Porphyromonas in periodontitis-associated with DM, (generally associated with more severe disease), and lower abundance of Streptococcus in this group (generally associated with less severe disease)]. The authors concluded that there were differences in the microflora between these two groups. Nevertheless, these differences are subtle, and do not change the current paradigm that focuses on the importance of enhanced inflammation underlying the association of DM and periodontitis.
An interesting study focused on measures of clinical inflammation in patients with DM and periodontitis.15Takeda K, Mizutani K, Minami I, et al. Association of periodontal pocket area with type 2 diabetes and obesity: a cross-sectional study. BMJ Open Diabetes Res Care. 2021;9(1) https://www.ncbi.nlm.nih.gov/pubmed/33879517. The surface area of inflamed pocket epithelium (PISA = periodontal inflamed surface area) and the epithelial surface area (PESA = periodontal epithelial surface area) were compared to several parameters associated with DM (HbA1c, fasting plasma glucose) and obesity (body mass index, visceral fat area). PISA was positively associated with both HbA1c and FPG, but not measures of weight. This study confirmed an association of clinical parameters, specifically periodontal tissue inflammation, with surrogate markers of metabolic control. This is additional evidence of the important role of enhanced inflammation in the association of DM and periodontitis.
Lastly, two recent systematic reviews have examined the relationship of periodontitis and microangiopathic complications of DM. Examining the literature on the relationship of periodontitis and retinopathy16Alvarenga MOP, Miranda GHN, Ferreira RO, et al. Association between diabetic retinopathy and periodontitis-a systematic review. Front Public Health. 2020;8:550614 https://www.ncbi.nlm.nih.gov/pubmed/33490007., five studies that were cross-sectional in design were included. There was a significant relationship between periodontitis severity and the severity of retinopathy, which was notable for persons who were not obese (odds ratio of 2.2). Another review assessed the literature for the relationship between periodontitis and microangiopathy (all types), retinopathy, nephropathy and neuropathy.17Zhang X, Wang M, Wang X, et al. Relationship between periodontitis and microangiopathy in type 2 diabetes mellitus: a meta-analysis. J Periodontal Res. 2021 https://www.ncbi.nlm.nih.gov/pubmed/34254680. Thirteen studies were included and found significant associations for the first three (odds ratios of 2.43, 4.33 and 1.75, respectively), but not neuropathy (odds ratio of 0.99). These associations were confirmed with further subgroup analyses. Since these studies were cross-sectional in nature, the temporal relationship of the development of periodontitis, and microangiopathy associated with type 2 DM, could not be determined.
These recent publications, both reviews and original research, strengthen our understanding of the importance of periodontal disease as a complication of DM, and how periodontal disease can affect metabolic control in patients with DM. These reports
- Further support the importance of DM as a risk factor for periodontitis.
- Provide additional supporting data that conservative periodontal treatment can improve glycemic control.
- Emphasize the critical role of inflammation in the bi-directional relationship of DM and periodontitis.
Given the high prevalence of DM in the population (approximately 13% of adults), the bidirectional association of DM and periodontal disease should be very familiar to all oral health care providers. Evaluation of every patient seen in the dental office begins with a review of the medical history, and a history of DM, or identification of risk factors for DM, should trigger a closer evaluation of the patient’s medical status. Conversely, the presence of moderate to advanced periodontitis that cannot be explained by the presence of local factors (i.e., accumulation of biofilm) should include an evaluation for DM.
Endodontic Complications:
The relationship of DM and periodontal disease has been closely linked to the exuberant inflammation response that characterizes DM. This results in a more intense local inflammatory response in the periodontium. Further, periodontal inflammation can contribute to the systemic inflammatory burden. In addition to periodontal disease, endodontic infections represent another oral disorder that may be modified when DM is present. Two recent systematic reviews, one with a meta-analysis, have examined this relationship. The systematic review with a meta-analysis examined the number of extracted teeth which received endodontic treatment. The meta-analysis revealed an odds ratio of 2.44, indicating that patients with DM had a significantly greater prevalence of endodontically-treated teeth that were extracted.18Cabanillas-Balsera D, Martin-Gonzalez J, Montero-Miralles P, et al. Association between diabetes and nonretention of root filled teeth: a systematic review and meta-analysis. Int Endod J. 2019;52(3):297-306 https://www.ncbi.nlm.nih.gov/pubmed/30168851. A subsequent systematic review evaluated 10 studies, and the authors concluded that type 2 DM is associated with an increased risk of developing endodontic lesions. This finding was related to several factors including a reduced healing response associated with DM. 19Rios-Osorio N, Munoz-Alvear HD, Montoya Canon S, et al. Association between type 2 diabetes mellitus and the evolution of endodontic pathology. Quintessence Int. 2020;51(2):100-7 https://www.ncbi.nlm.nih.gov/pubmed/31942571.
Healing Following Endodontic Treatment
A clinical study which examined dental records of patients receiving root canal treatment during a 3-year period (2008-2011) was conducted to assess success of root canal treatment in patients with DM.20Laukkanen E, Vehkalahti MM, Kotiranta AK. Impact of systemic diseases and tooth-based factors on outcome of root canal treatment. Int Endod J. 2019;52(10):1417-26 https://www.ncbi.nlm.nih.gov/pubmed/31074887. Patients without systemic disease, with cardiovascular disease, and with DM were followed for 6 to 71 months (mean of 22.7 months). For patients without systemic disease, the success rate of endodontic treatment was 85.6%. However, the success rate of treatment in patients with DM was significantly lower (73.2%). The lower success rate was not seen for patients with cardiovascular disease. Regression analysis revealed that tooth-specific factors (i.e., the quality of the root filling, the amount of alveolar bone loss) were more important than DM status in determining the clinical outcome.
A study of a large cohort of patients from a hospital network21Yip N, Liu C, Wu D, et al. The association of apical periodontitis and type 2 diabetes mellitus: a large hospital network cross-sectional case-controlled study. J Am Dent Assoc. 2021;152(6):434-43 https://www.ncbi.nlm.nih.gov/pubmed/33795142. examined the relationship of apical periodontitis and type 2 DM. The subset of data included 7749 patients that had been diagnosed with apical periodontitis, and 7749 control patients who did not have apical periodontitis. The groups were matched by age, sex, and race. Regression analysis examined the relationship of DM and apical periodontitis, controlling for demographic variables, systemic disease, DM (HbA1c), periodontitis, BMI, smoking and medication use (metformin and statins). They determined that type 2 DM was associated with a higher prevalence of apical periodontitis (odds ratio = 2.05). Using metformin (odds ratio = 0.82) and statins (odds ratio = 0.70) was associated with a lower prevalence of apical periodontitis, but if the HbA1c was 8.0% or greater, prevalence was higher (odds ratio = 2.46). The authors concluded that type 2 DM, especially if there was poor metabolic control, was linked to greater prevalence of apical periodontitis.
Similarly, a systematic review and meta-analysis examined apical healing associated with endodontically-treated teeth in patients with DM, as compared to controls.22Gupta A, Aggarwal V, Mehta N, et al. Diabetes mellitus and the healing of periapical lesions in root filled teeth: a systematic review and meta-analysis. Int Endod J. 2020;53(11):1472-84 https://www.ncbi.nlm.nih.gov/pubmed/32654191. A total of 10 studies were included, and they considered both observational and clinical studies. The odds ratios for reduced healing in these two types of studies were 1.42 and 6.36, respectively, confirming altered healing of endodontically treated teeth in the presence of DM.
However, not all recent findings support association of DM and endodontic complications. A study examined the level of glycemic control in relation to the occurrence of endodontically treated teeth and periapical lesions in persons with diabetes.23Perez-Losada FL, Lopez-Lopez J, Martin-Gonzalez J, et al. Apical periodontitis and glycemic control in type 2 diabetic patients: cross-sectional study. J Clin Exp Dent. 2020;12(10):e964-e71 https://www.ncbi.nlm.nih.gov/pubmed/33154799. Radiographic records were analyzed in a cross-sectional design. Only patients with DM were included but were differentiated based on level of glycemic control (HbA1c of <6.5%, or > 6.5%). For the entire cohort, the mean HbA1c was 7.0% (mean of 6.0% for the well-controlled group and 7.8% for the poorly controlled group). The authors did not find any correlation between glycemic control and the presence apical periodontitis, the presence of a root canal-filled tooth or the presence of root canal-filled teeth with a periapical lesion. These data should be interpreted with caution, as the mean concentration of HbA1c in this population was 7.0%. From the clinical perspective, this concentration of HbA1c is considered adequate metabolic control. Nevertheless, taken in total, the findings suggest that the relationship of DM and endodontic complications is not as clear as it is for DM and periodontitis. That is not surprising, as endodontic complications of DM can be considered indirectly modified by DM, as such lesions would follow the development of a carious lesion, which could be influenced by a variety of other variables (i.e., diet, salivary flow).
Understanding the DM - Endodontic Relationship
Other recent studies of the relationship of DM and endodontic complications have focused on the microbiology of endodontic lesions in persons with DM. A small study identified a much higher percentage of samples that were positive for Candida albicans from both the periodontal pockets and the root canals from persons with DM vas compared to persons without the disease.24Gomes CC, Guimaraes LS, Pinto LCC, et al. Investigations of the prevalence and virulence of Candida albicans in periodontal and endodontic lesions in diabetic and normoglycemic patients. J Appl Oral Sci. 2017;25(3):274-81 https://www.ncbi.nlm.nih.gov/pubmed/28678946. The fungal organisms collected from patients with DM demonstrated greater virulence than the fungal organisms from persons without the disease.
Another small study examined the presence of important periodontal pathogens (Porphyromonas gingivalis, Prevotella intermedia) and an endodontic pathogen (Enterococcus faecalis) from both periodontal pockets and root canal samples.25Nair RR, Nayak M, Prasada LK, et al. PCR-based detection of three anaerobic bacteria associated with endodontic-periodontic lesions in type-2 diabetic and nondiabetic subjects. J Conserv Dent. 2019;22(5):430-5 https://www.ncbi.nlm.nih.gov/pubmed/33082657. The percentage of these 3 bacterial species in the root canal of patients with DM was 73% for E. faecalis, 70% P. gingivalis and 36% P. intermedia. In contrast, these percentages from the non-DM patients were 53%, 43% and 23%. In the periodontal samples from patients with DM, these percentages were 30%, 73% and 50%, versus almost equal percentages in persons without DM (30%, 37%, 33%). The authors suggested that the high percentage of periodontal and endodontic samples from patients with DM with P. gingivalis (73% and 70%) suggest the possibility of periodontal-endodontic communication.
These recent studies are evidence of continued interest in how DM can influence the development and treatment outcomes for endodontic complications. This represents another aspect of the increasingly complex relationship between DM and the oral cavity. In the larger sense, DM has been shown to have a wide variety of effects on the oral cavity, indicating the need for oral health care providers to be part of care management for persons with DM. In the second essay, tooth loss and implant complications will be discussed. In the third essay, recent findings regarding less common oral complications of DM will be reviewed, as well as identification of dysglycemia in the dental office, and studies suggesting that provision of preventive dental care for persons with DM can improve health outcomes.
References
- 1.Centers for Disease Control and Prevention. National Diabetes Statistics Report, 2020. Atlanta, GA: Centers for Disease Control and Prevention, U.S. Dept of Health and Human Services; 2020.
- 2.International Diabetes Federation. IDF Diabetes Atlas, 9th edn. Diabetes Facts and Figures. Brussels, Belgium: 2019. Available at: https://www.diabetesatlas.org.
- 3.Fang M, Wang D, Coresh J, et al. Trends in diabetes treatment and control in U.S. adults, 1999-2018. N Engl J Med. 2021;384(23):2219-28 https://www.ncbi.nlm.nih.gov/pubmed/34107181.
- 4.Verhulst MJL, Loos BG, Gerdes VEA, et al. Evaluating all potential oral complications of diabetes mellitus. Front Endocrinol (Lausanne). 2019;10:56 https://www.ncbi.nlm.nih.gov/pubmed/30962800.
- 5.Stohr J, Barbaresko J, Neuenschwander M, et al. Bidirectional association between periodontal disease and diabetes mellitus: a systematic review and meta-analysis of cohort studies. Sci Rep. 2021;11(1):13686 https://www.ncbi.nlm.nih.gov/pubmed/34211029.
- 6.Zhang S, Philips KH, Moss K, et al. Periodontitis and Risk of Diabetes in the Atherosclerosis Risk In Communities (ARIC) Study: A BMI-modified association. J Clin Endocrinol Metab. 2021 https://www.ncbi.nlm.nih.gov/pubmed/33982107.
- 7.Dicembrini I, Serni L, Monami M, et al. Type 1 diabetes and periodontitis: prevalence and periodontal destruction-a systematic review. Acta Diabetol. 2020;57(12):1405-12 https://www.ncbi.nlm.nih.gov/pubmed/32318875.
- 8.Zainal Abidin Z, Zainuren ZA, Noor E, et al. Periodontal health status of children and adolescents with diabetes mellitus: a systematic review and meta-analysis. Aust Dent J. 2021 https://www.ncbi.nlm.nih.gov/pubmed/33864280.
- 9.Chakraborty P, Mukhopadhyay P, Bhattacharjee K, et al. Periodontal disease in type 1 diabetes mellitus: influence of pubertal stage and glycemic control. Endocr Pract. 2021;27(8):765-8 https://www.ncbi.nlm.nih.gov/pubmed/33486090.
- 10.Heji ES, Bukhari AA, Bahammam MA, et al. Periodontal disease as a predictor of undiagnosed diabetes or prediabetes in dental patients. Eur J Dent. 2021;15(2):216-21 https://www.ncbi.nlm.nih.gov/pubmed/33285572.
- 11.Rapone B, Ferrara E, Corsalini M, et al. Inflammatory status and glycemic control level of patients with type 2 diabetes and periodontitis: a randomized clinical trial. Int J Environ Res Public Health. 2021;18(6) https://www.ncbi.nlm.nih.gov/pubmed/33804123.
- 12.Chen YF, Zhan Q, Wu CZ, et al. Baseline HbA1c level influences the effect of periodontal therapy on glycemic control in people with type 2 diabetes and periodontitis: a systematic review on randomized controlled trails. Diabetes Ther. 2021;12(5):1249-78 https://www.ncbi.nlm.nih.gov/pubmed/33481189.
- 13.Cirelli T, Nepomuceno R, Goveia JM, et al. Association of type 2 diabetes mellitus and periodontal disease susceptibility with genome-wide association-identified risk variants in a Southeastern Brazilian population. Clin Oral Investig. 2021;25(6):3873-92 https://www.ncbi.nlm.nih.gov/pubmed/33392810.
- 14.Balmasova IP, Olekhnovich EI, Klimina KM, et al. Drift of the subgingival periodontal microbiome during chronic periodontitis in type 2 diabetes mellitus patients. Pathogens. 2021;10(5) https://www.ncbi.nlm.nih.gov/pubmed/33922308.
- 15.Takeda K, Mizutani K, Minami I, et al. Association of periodontal pocket area with type 2 diabetes and obesity: a cross-sectional study. BMJ Open Diabetes Res Care. 2021;9(1) https://www.ncbi.nlm.nih.gov/pubmed/33879517.
- 16.Alvarenga MOP, Miranda GHN, Ferreira RO, et al. Association between diabetic retinopathy and periodontitis-a systematic review. Front Public Health. 2020;8:550614 https://www.ncbi.nlm.nih.gov/pubmed/33490007.
- 17.Zhang X, Wang M, Wang X, et al. Relationship between periodontitis and microangiopathy in type 2 diabetes mellitus: a meta-analysis. J Periodontal Res. 2021 https://www.ncbi.nlm.nih.gov/pubmed/34254680.
- 18.Cabanillas-Balsera D, Martin-Gonzalez J, Montero-Miralles P, et al. Association between diabetes and nonretention of root filled teeth: a systematic review and meta-analysis. Int Endod J. 2019;52(3):297-306 https://www.ncbi.nlm.nih.gov/pubmed/30168851.
- 19.Rios-Osorio N, Munoz-Alvear HD, Montoya Canon S, et al. Association between type 2 diabetes mellitus and the evolution of endodontic pathology. Quintessence Int. 2020;51(2):100-7 https://www.ncbi.nlm.nih.gov/pubmed/31942571.
- 20.Laukkanen E, Vehkalahti MM, Kotiranta AK. Impact of systemic diseases and tooth-based factors on outcome of root canal treatment. Int Endod J. 2019;52(10):1417-26 https://www.ncbi.nlm.nih.gov/pubmed/31074887.
- 21.Yip N, Liu C, Wu D, et al. The association of apical periodontitis and type 2 diabetes mellitus: a large hospital network cross-sectional case-controlled study. J Am Dent Assoc. 2021;152(6):434-43 https://www.ncbi.nlm.nih.gov/pubmed/33795142.
- 22.Gupta A, Aggarwal V, Mehta N, et al. Diabetes mellitus and the healing of periapical lesions in root filled teeth: a systematic review and meta-analysis. Int Endod J. 2020;53(11):1472-84 https://www.ncbi.nlm.nih.gov/pubmed/32654191.
- 23.Perez-Losada FL, Lopez-Lopez J, Martin-Gonzalez J, et al. Apical periodontitis and glycemic control in type 2 diabetic patients: cross-sectional study. J Clin Exp Dent. 2020;12(10):e964-e71 https://www.ncbi.nlm.nih.gov/pubmed/33154799.
- 24.Gomes CC, Guimaraes LS, Pinto LCC, et al. Investigations of the prevalence and virulence of Candida albicans in periodontal and endodontic lesions in diabetic and normoglycemic patients. J Appl Oral Sci. 2017;25(3):274-81 https://www.ncbi.nlm.nih.gov/pubmed/28678946.
- 25.Nair RR, Nayak M, Prasada LK, et al. PCR-based detection of three anaerobic bacteria associated with endodontic-periodontic lesions in type-2 diabetic and nondiabetic subjects. J Conserv Dent. 2019;22(5):430-5 https://www.ncbi.nlm.nih.gov/pubmed/33082657.
Figure 1. Age-adjusted estimated prevalence of diagnosed diabetes by race/ethnicity group and sex for adults aged 18 years or older, United States, 2017-2018. Source: reference 1.
Note: Error bars represent upper and lower bounds of the 95% confidence interval.
Data sources: 2017-2018 National Health Interview Survey; 2017 Indian Health Service National Data Warehouse (for American Indian/ Alaska Native group only).
Figure 2. Global prevalence of diabetes in adults (20-79 years) in 2019, 2030 and 2045. Source: reference 2.
Figure 3. A patient with previously undiagnosed DM who was seen for periodontal evaluation. The palatal tissues display evidence of periodontal abscesses (suppuration) and granulomatous change of the marginal tissues (evidence of poor wound healing).